Renal Intra-Arterial Delivery of MSC to Ischemic Porcine Kidneys: Retainment, Localization and Survival
Jesus M. Sierra Parraga1, Marco Eijken2, Christine Andersen2, Cyril Moers3, Rutger Ploeg4, Bjarne Møller5, Carla C. Baan1, Martin J. Hoogduijn1, Bente Jespersen6.
1Nephrology and Transplantation, Internal Medicine, Erasmus MC, Rotterdam, Netherlands; 2Institute of Clinical Medicine, Aarhus University, Aarhus, Denmark; 3Organ Donation and Transplantation, University Medical Center Groningen, Groningen, Netherlands; 4Nuffield Department of Surgical Sciences and Oxford Biomedical Research Centre , University of Oxford, Oxford, United Kingdom; 5Department of Clinical Immunology, Aarhus University Hospital, Aarhus, Denmark; 6Department of Renal Medicine, Aarhus University Hospital, Aarhus, Denmark
MePEP consortium.
Donor organ shortage is one of the main problems in kidney transplantation. The increase in the available donor kidney pool comes with the use of expanded criteria donor organs, leading to a poorer outcome for the grafts and the patients. Therefore, a number of techniques are arising to improve the organ quality, such as machine perfusion in combination with cell therapy. Mesenchymal stem cells (MSC) have tissue regenerative capacities exerted by paracrine secretion of different cytokines and physical interaction with surrounding cells.
Porcine MSC from adipose tissue were isolated and grown to carry out the experiments. A model of unilateral ischemia-reperfusion injury by clamping the renal artery for 1 hour was established. 10 million MSC were fluorescently labelled with Qtracker beads and infused directly through the renal artery during 10 minutes. Fluorescent MSC were detected using flow cytometry Before, during and after the infusion, blood samples were taken from the renal vein to assess if MSC were leaving the kidney. After 30 minutes or 8 hours both kidneys were retrieved and biopsies were collected from multiple sites. These biopsies were mechanically disrupted and enzymatically digested to a single cell suspension and analyzed by flow cytometry. Slices from paraffin-embedded biopsies were stained by hematoxylin and eosin and analyzed by light and fluorescence microscopy to identify the exact localization of the administered MSC.
Flow cytometric analyses of blood samples allowed us to observe MSC leaving the kidney (up to 2000 MSC/ml blood) during the infusion procedure, after which very low fluorescent cells were detected in renal venous blood (300 MSC/ml). Arterial blood was also analyzed but no MSC was identified. Flow cytometric analysis of dissociated renal cortex and medulla revealed that most MSC were present in renal cortex (8000-20000 MSC/gram of cortex) but a number of individual MSC were detected in the medulla (1000-3000 MSC/gram of medulla). From these MSC inside renal tissue, 80% were alive. Lung tissue was also analyzed and shown to contain a small population of MSC (200-400 MSC/ gram). Microscopy data validated these results. MSC were detected in the renal cortex, mainly inside the glomeruli but individual MSC were found also in tubuli, vessels and medulla.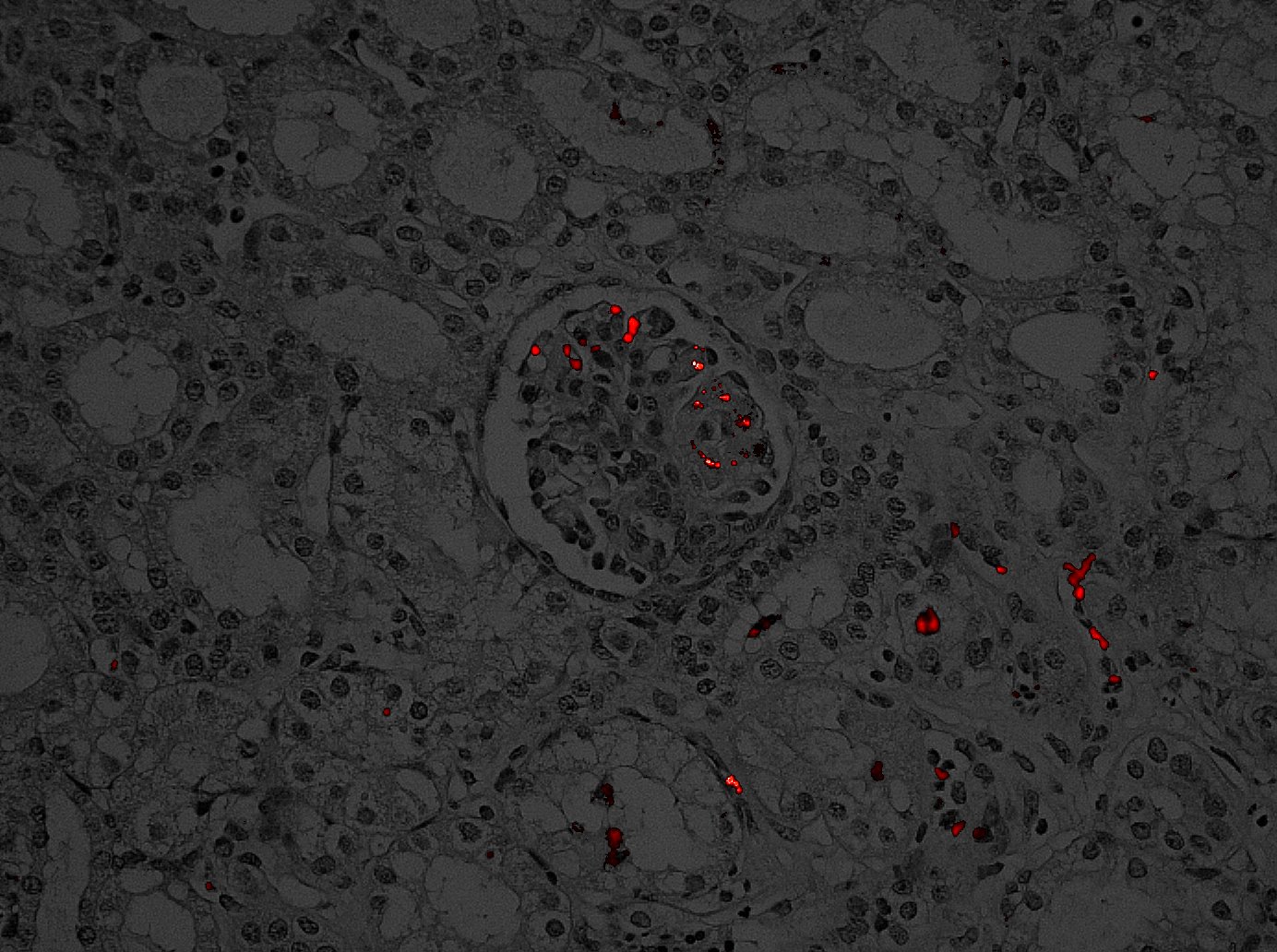
Delivery of MSC through the renal artery can successfully deliver MSC throughout the kidney. MSC were retained particularly in glomeruli for at least 8 hours. We were able to estimate the relative retainment and localization of MSC in the kidney. These studies will pave the way for survival studies in this large animal model aiming for future clinical studies with targeted delivery of MSC in renal disease, eg. in transplantation of high risk donor organs.
Lundbeck Foundation under grant application no. R198-2015-184..
