Donor’s Graft Ex Vivo T Cell Depletion with Fludarabine Reduces GvHD Signs and Improves Survival after Intestine Transplantation
Maria Vela1, Pablo Stringa1, Pablo González-Navarro1, Tania S Rubio2, Jaime Valentín1, Alba Cano-Rodríguez1, Eduardo López-Granados3, Eduardo López-Collazo1, Francisco Hernández-Oliveros4, Antonio Pérez-Martínez1.
1Innate Immunity, Hospital La Paz Institute for Health Research (IdiPAZ), Madrid, Spain; 2Laboratory Medicine, La Paz University Hospital, Madrid, Spain; 3Immunology, Hospital La Paz Institute for Health Research (IdiPAZ), Madrid, Spain; 4Pediatric Transplant Surgery, La Paz University Hospital, Madrid, Spain
Introduction: Intestine passenger T leukocytes are responsible of graft versus host disease (GvHD) in intestine transplantation. These lymphocytes are known to have inferior tolerogenic qualities compared with leukocytes in the liver, other solid organs and bone marrow. Fludarabine is a routinely used anti neoplasic agent with high cytotoxicity against T cells. We hypothesized that ex vivo fludarabine treatment of the bowel graft could diminish the risk of GvHD and improve post transplant overall survival.
Methods: We performed isolated heterotopic small bowel transplantations from Lewis (LEW) to Brown Norway (BN) rat strains. The grafts of one of the experimental groups were imbided and sealed in Celsior preserving solution with 1000 µM fludarabine during surgery (1 h), before its implantation into recipient animals.
We compared a group of untreated (n = 7) vs a group of fludarabine-treated bowel recipients (n = 5). The mixed hematopoietic chimerism was determined by flow cytometry using strain-specific anti HLA antibodies. Clinical signs of GvHD as well as post-transplant overall survival were also monitored.
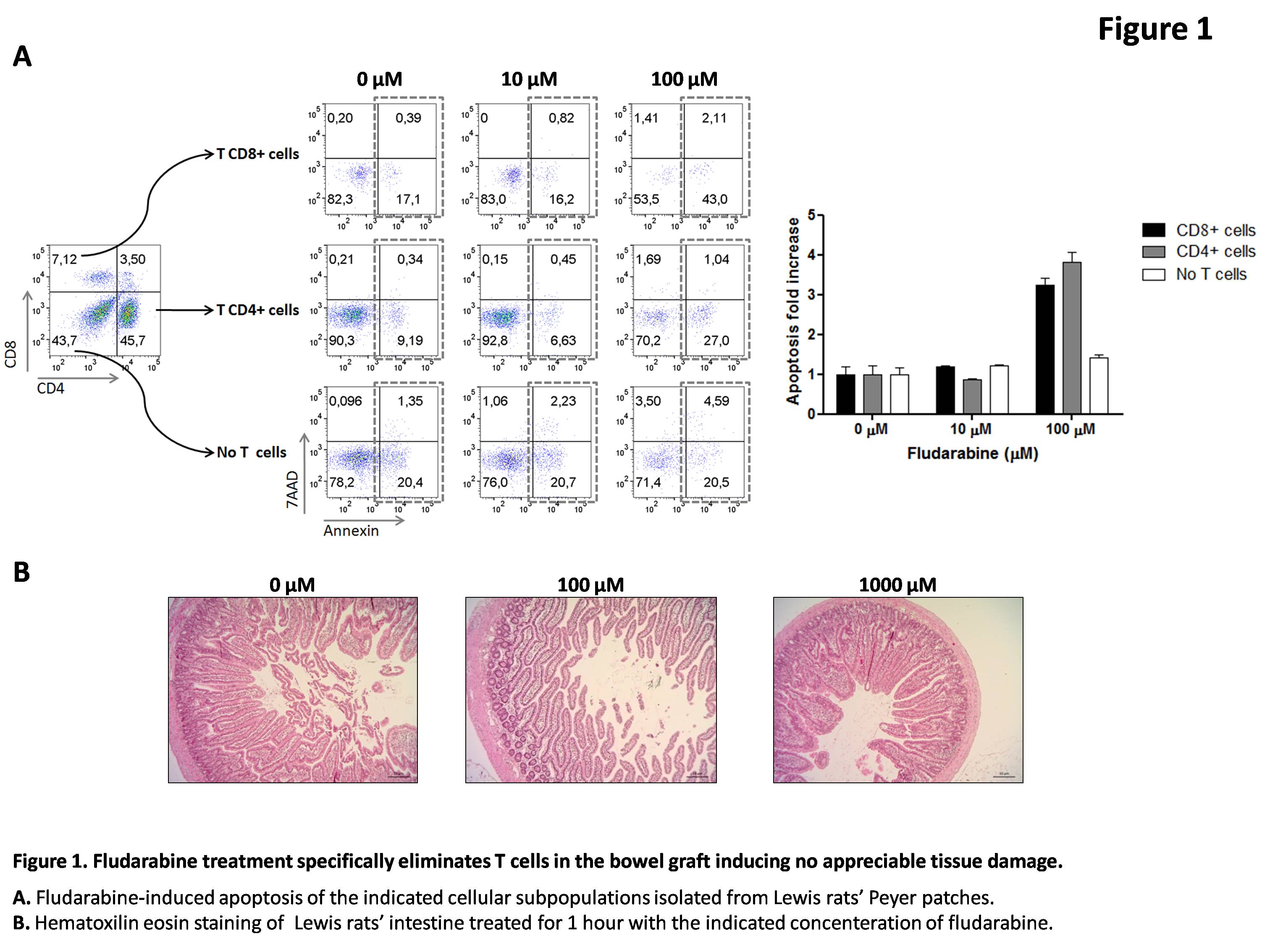
Results: One hour fludarabine treatment of the bowel grafts induced specific apoptosis of its passenger T cells at concentrations from 100 μM while no histological signs of intestinal tissue alterations were observed after 1000 μM fludarabine treatment (Figure 1).
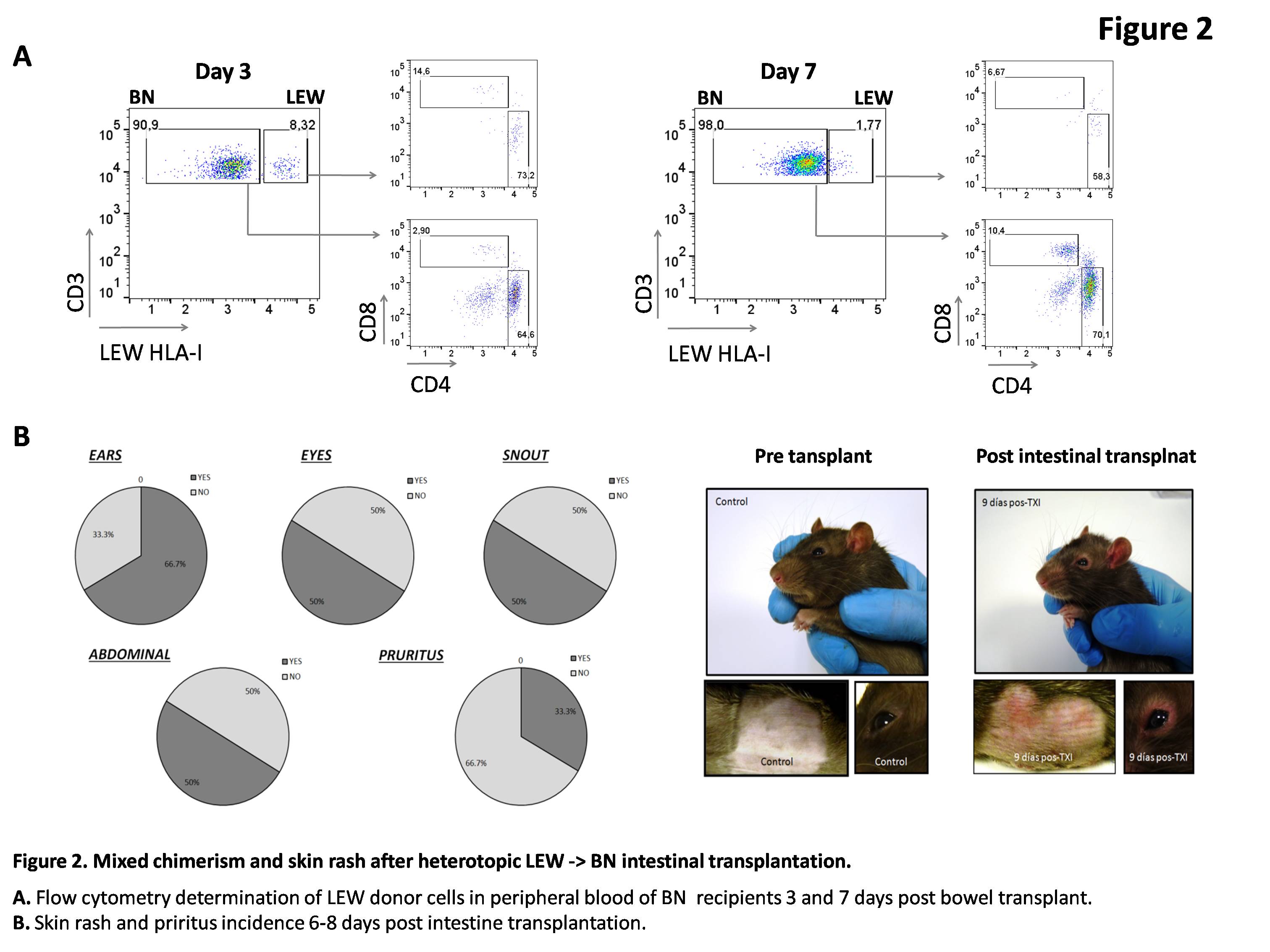
After heterotopic LEW -> BN intestinal transplantation, untreated intestine recipients showed GvHD signs from the fourth day post transplant (n = 7). These symptoms include: rash (n = 4), weight loss (n = 3), piloerection (n = 2) and diarrhea (n = 1). The chimerism or percentage of donor’s lymphocytes in the recipient rat, determined in peripheral blood, reached 6.61% (range 1.7-10.8%) at day 3 post transplantation and 2.56% (range 0-9.8%) at day 7 post transplantation (Figure 2).
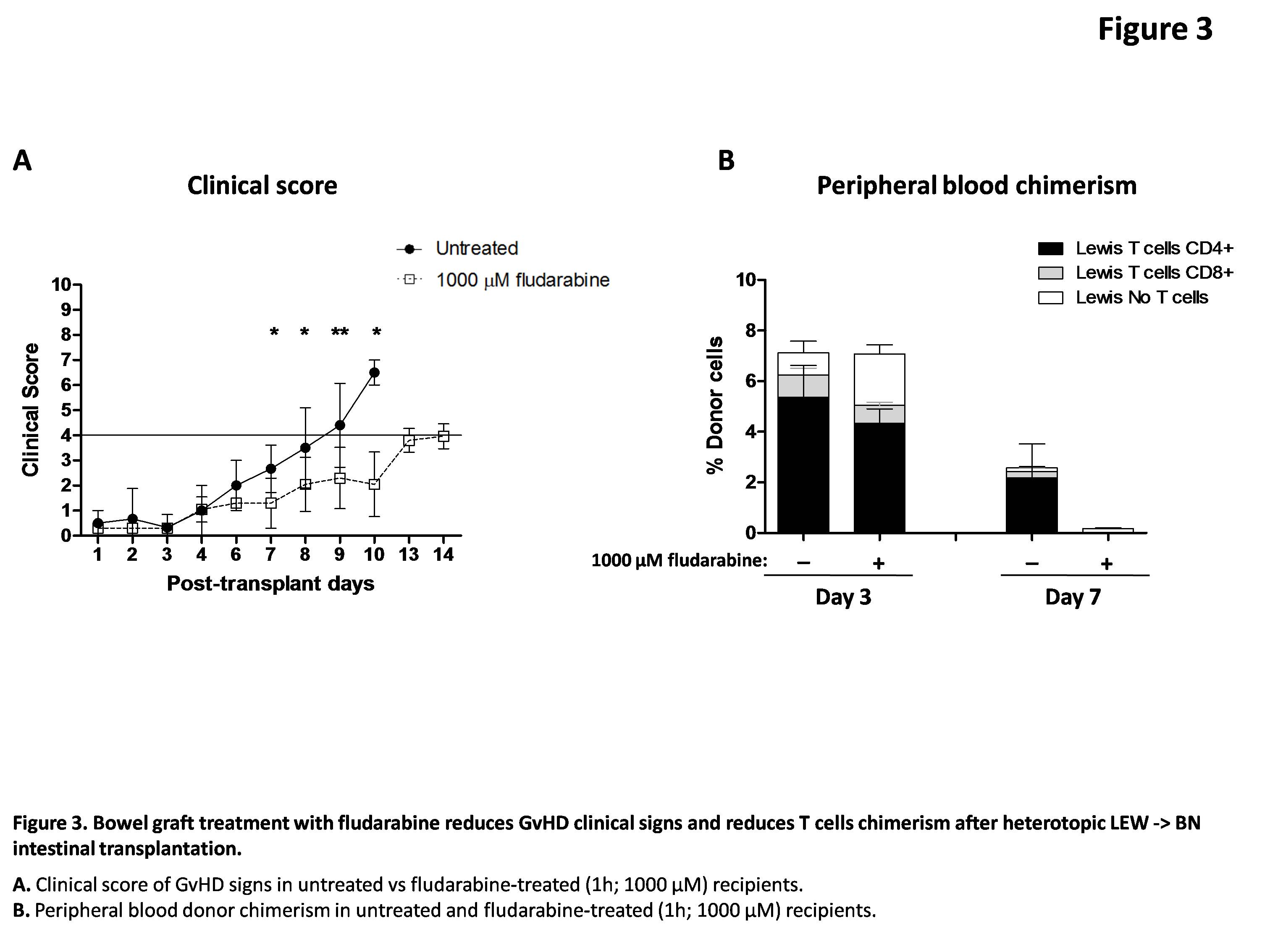
Rats transplanted with a fludarabine-conditioned intestine showed statistically signifcant later and milder clinical signs of GvHD (Figure 3A). Additionally, fludarabine treatment reduced total donor cells chimerism at day 7 and the percentage of chimeric T cells at days 3 and 7 post transplant (Figure 3B).
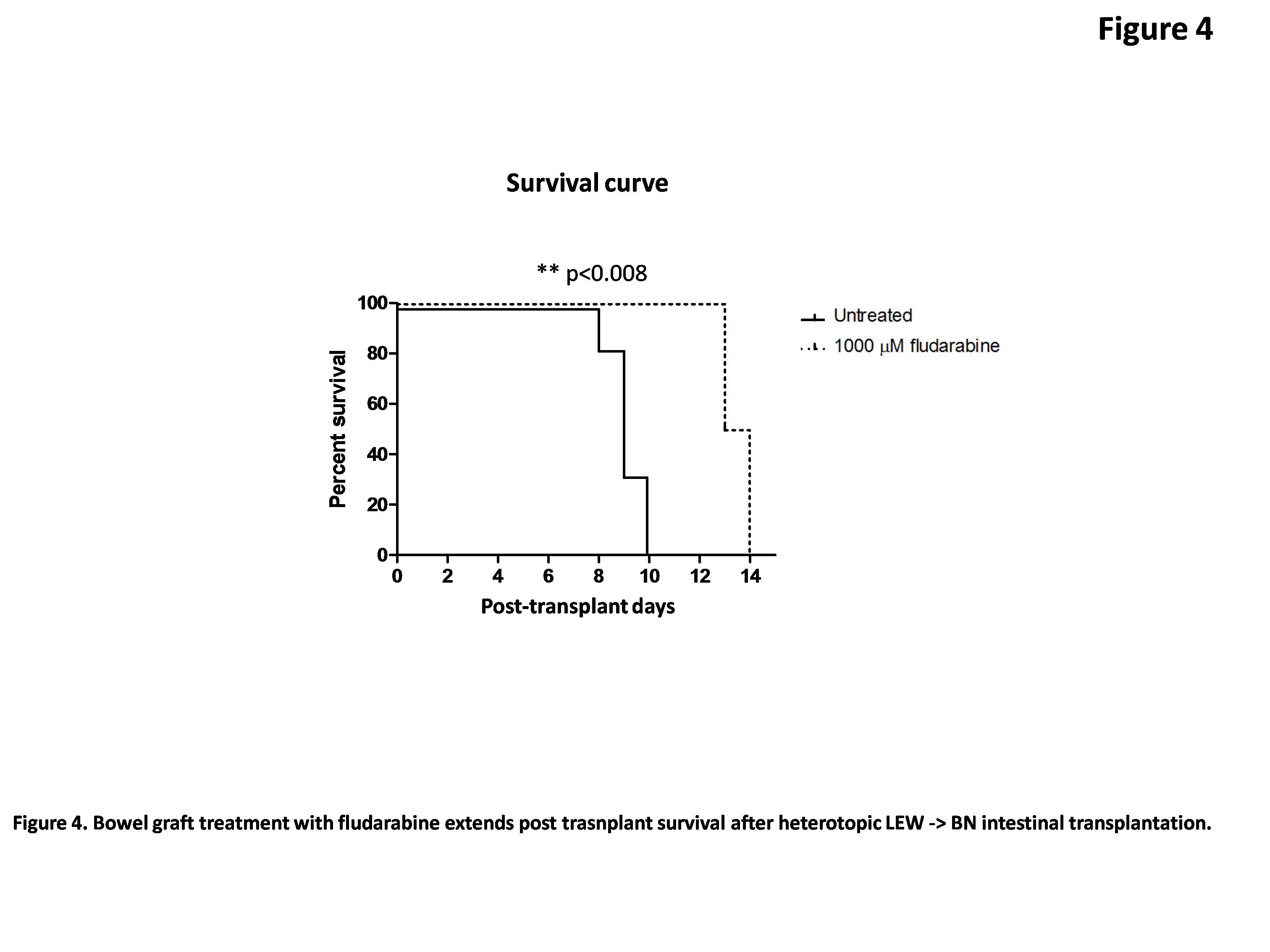
Untreated bowel grafts recipients died within 9.2 days ± 0.3 days while fludarabine-treated graft recipients showed prolonged survival (13.5 days ± 0.3 days) (Figure 4).
Conclusions: Graft immunosupresion with fludarabine during surgical procedure protects bowel recipients of GvHD risk and improves post transplant overall survival.
TrasplantChild European Reference Network.
