B-Cell Depletion Prevents Humoral Sensitization after Pig-to-Baboon Lung Xenotransplantation
Agnes Azimzadeh1, Wessam Hassanein1, Lars Burdorf1, Wenji Sun1, Gheorghe Braileanu1, Christopher Laird1, Natalie A O'Neill1, Xiangfei Cheng1, David l Ayares2, Richard N Pierson III1.
1Surgery, University of Maryland Baltimore School of Medicine, Baltimore, MD, United States; 2Revivicor, Blacksburg, VA, United States
Background: The survival of lung xenografts is currently limited by graft injury consistent with immune and inflammatory mediated injury. GalTKO pigs expressing several additional human transgenes are evaluated to control innate immunity, coagulation dysregulation and inflammatory processes. However, induced antibodies directed against non-gal antigens (αNGAb) likely play an important role in rejection of lung and other xenografts. Here, we evaluate whether B-cell depletion suppresses antibody responses and impacts the outcome of pig-to-baboon lung xenografts in the context of an established costimulation blockade-based immunosuppressive regimen.
Materials and Methods: Seven baboons received an orthotopic left lung xenograft from GalTKO.hCD46 pigs expressing several additional human transgenes and immunosuppression based on T-cell depletion (ATG) and αCD40 costimulation blockade, five with additional B-cell depletion using αCD20 Ab, modeled after Mohiuddin et al. (Nature Communications, 2016). αNGAb levels were measured in baboon serum by adhesion to GalTKO pig aortic endothelial cells using flow cytometry.
Results: Peripheral B-cell counts decreased dramatically after treatment with αCD20 Ab (from~2000 to 33±58 CD19+ cells/ul) and remained low during study interval (Figure 1A, red curves). Shortly following lung transplantation, αNGAb IgM levels decreased in both groups (Figure 1B), presumably reflecting adsorption to the xenograft. A strong rebound in IgM levels (>1000 fold baseline levels) was detected one week after transplantation with baseline immunosuppression (black curves), but was dramatically attenuated by B-cell depletion (Figure 1B, red curves). However, IgM levels increased slightly above baseline levels in two animals surviving to day 7. αNGAb IgG levels followed the same general trend albeit with much lower antibody levels (Figure 1C). All lungs exhibited vascular barrier function injury (alveolar flooding and hemorrhage) within 6-9 days. 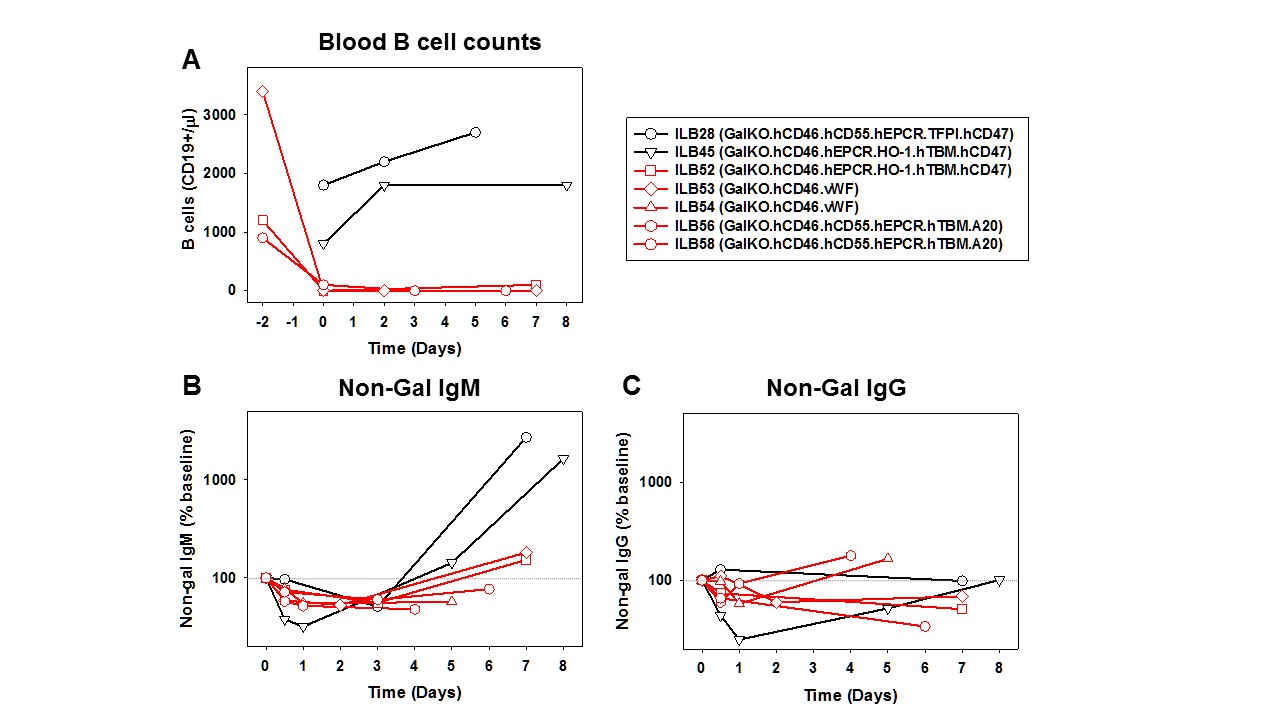
Conclusion: CD20-based B-cell depletion suppresses sensitized humoral responses to non-gal antigens, as seen in other organ xeno-models. However, aNGAb return to pre-transplant levels within a week independent of B cell depletion. Inhibiting induced αNGAb elaboration is not sufficient to prevent early lung xenograft failure, suggesting that preformed antibodies insensitive to B cell depletion and/or antibody-independent mechanisms are critical to the pathogenesis of lung xenograft injury relative to other organ xenografts.
